Description
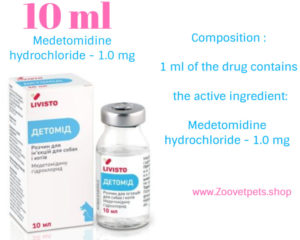
Description :
Transparent colorless sterile aqueous solution.
Composition :
1 ml of the drug contains
the active ingredient:
Medetomidine hydrochloride – 1.0 mg
(Medetomidine equivalent 0.85 mg).
Excipients: methyl parahydroxybenzoate (E218), propyl parahydroxybenzoate (E216), sodium chloride, water for injection.
Pharmacological properties :
ATC vet classification code: QN05CM- Other hypnotic and sedative veterinary drugs. QN05CM91- Medetomidine.
Medetomidine is a selective alpha-2-adrenoreceptor agonist that acts both centrally (general action) and peripherally (local action), both presynaptically and postsynaptically, reduces consciousness and increases pain threshold. Activation of these receptors causes a decrease in norepinephrine release in the central nervous system, manifested by sedation, analgesia, and bradycardia. At the peripheral level, medetomidine causes vasoconstriction through stimulation of postsynaptic alpha-2-adrenoreceptors, resulting in temporary hypertension. Blood pressure returns to normal (sometimes slightly lower than normal) in 1-2 hours. Respiratory rate may temporarily slow down.
The duration and depth of sedation and analgesia depend on the dose :
In addition to its sedative, analgesic and myorelaxing properties, medetomidine also has hypothermic and mydriatic effects (dilates the pupils), inhibits salivation and slows intestinal peristalsis.
After intramuscular administration, medetomidine is rapidly and almost completely absorbed from the injection site. Maximum concentration in blood plasma is observed 15-20 minutes after injection, the elimination half-life is 1.2 hours in dogs and 1.5 hours in cats. Medetomidine is predominantly oxidized in the liver, while a small amount is methylated in the kidneys. The metabolites are excreted mainly in the urine.
Applications :
Dogs and cats: for sedation and analgesia associated with clinical examinations and procedures, minor surgical examinations, and as a premedication before general anesthesia.
Dosage :
Dogs:
Intramuscularly or intravenously.
For sedation, the dose is 10-80 mcg of medetomidine hydrochloride per 2.2lb (1 kg) body weight intravenously or intramuscularly.
Maximum effect is achieved in 15-20 minutes. Clinical effect depends on the dose and lasts from 30 to 180 minutes.
For premedication, the drug should be administered in a dose of 10-40 mcg of medetomidine hydrochloride per 2.2b (1 kg) body weight, which is equivalent to 0.1-0.4 ml per 22lb (10 kg) body weight. The exact dose depends on the combination of drugs used and the dose(s) of the other drug(s).
In addition, the dose should be adjusted with the type of surgical intervention, the duration of the procedure, the temperament, and the body weight of the dog. Premedication with medetomidine will greatly reduce the required dose of induction agent and reduce the need for volatile anesthetics to maintain anesthesia. All anesthetic agents used for induction or maintenance of anesthesia should be administered at the appropriate dose. You should read the postcard insert of other veterinary medications before using any combination.
Cats:
Intramuscular, intravenous, or subcutaneous
For sedation, the drug is used in a dose of 50-150 mcg of medetomidine hydrochloride per 1 kg body weight (0.05-0.15 ml per 2.2lb (1 kg) body weight).
For premedication in anesthesia the drug should be administered in a dose of 80 mcg of medetomidine hydrochloride per 2.2lb (1 kg) of body weight (0.08 ml of the drug per 2.2lb (1 kg) of body weight).
The rate of induction is slower when the subcutaneous route of administration is used.
An appropriate graduated syringe should be used to accurately administer the desired dose volume. This is especially important when small volumes are administered.
Contraindications :
Do not use in animals with severe cardiovascular, respiratory, hepatic or renal disease.
Do not use if there are mechanical disorders of the digestive tract (torsion of the stomach, hernia, obstruction of the esophagus).
Do not use in case of known hypersensitivity to the active substance or any of the excipients.
Do not use in animals with diabetes.
Do not administer to animals in a state of shock, emaciation or severe fatigue.
Do not administer to animals with eye problems, namely, high eye pressure.
Do not use simultaneously with sympathomimetic amines or sulfonamides and trimethoprim.
Cautions :
Side effects.
In very rare cases after drug administration bradycardia with AV blockade (1st and 2nd degree) and extrasystoles, coronary artery bypass grafting, reduced heart rate and increased arterial blood pressure (which subsequently returns to normal or slightly below normal) may occur.
Rarely, dogs and cats may experience vomiting 5-10 minutes after drug administration. Infrequently, repeated vomiting in cats may be observed.
Pulmonary edema, respiratory depression and cyanosis, increased diuresis, hypothermia, sensitivity to loud sounds, reversible hyperglycemia due to reduced insulin secretion, pain at the injection site and trembling of muscles may also be observed.
For cardiovascular and respiratory depression, assisted ventilation and oxygen administration may be prescribed. Atropine may increase heart rate
Dogs weighing less than 10 kg may be more likely to have the above adverse reactions.
Special cautions when using :
Medetomidine may cause respiratory depression, and manual ventilation and oxygen administration may be required in these cases.
A clinical examination of all animals should be performed prior to the use of veterinary drugs intended for sedation and/or general anesthesia.
When a veterinary drug is used for premedication, the dose of the anesthetic should be proportionately reduced and set according to the animal’s response. Special reservations and contraindications for the use of other veterinary drugs should be adhered to before any combination of anesthesia drugs is used.
High doses of medetomidine for large breeding dogs should be avoided. Care must be taken when combining medetomidine with other anesthetics or sedatives, as it has a marked potentiating effect on anesthetics.
Animals should be fed 12 hours before anesthesia.
Dogs and cats should be placed in a calm and quiet environment for the sedative effect to reach its maximum effect. This occurs in 10 to 15 minutes. No treatment or use of other medications should be started before maximum sedation is achieved.
Animals to which the drug is administered should be kept in a warm room both during treatment and during recovery.
The cornea of the eyes of animals to which the drug was administered should be moistened.
Animals should be calmed down before starting treatment.
If the dog or cat is ill or weak, they should be prescribed medetomidine only prior to administration and maintenance of general anesthesia based on a benefit/risk assessment.
Medetomidine should be used with caution in animals with cardiac disease, old or, animals with poor physical condition. Liver and renal function should be evaluated before use.
To reduce the recovery time after anesthesia or sedation, the effect of the veterinary drug may be reversed by administration of an alpha-2 adrenoreceptor antagonist such as atipamezole. Atipamezole does not alter the effects of ketamine. Alpha-2-adrenoceptor antagonists should not be administered earlier than 30-40 minutes after receipt of ketamine to avoid the muscle contraction that ketamine may cause.
Use during pregnancy, lactation, and egg production
The safety of the veterinary drug has not been established during pregnancy and lactation, so it should not be used.
Interaction with other drugs and other forms of interaction
Do not use simultaneously with sympathomimetic amines or sulfonamides and trimethoprim.
Caution should be exercised when administered concomitantly with agents that suppress central nervous system function – the effects of these drugs may be enhanced.
The effects of medetomidine may cause resistance when administering atipamezole.
Bradycardia can be partially prevented by prior administration (at least 5 minutes before) of an anticholinergic agent; however, administration of anticholinergic agents to treat bradycardia, simultaneously with medetomidine or after sedation with medetomidine, may result in adverse cardiovascular effects.
Special Cautions for Persons and Attendants
In case of accidental ingestion or accidental injection, seek medical advice immediately and show the postcard insert.
Never drive, as sedation or changes in blood pressure may occur!
Avoid contact with skin, eyes and mucous membranes!
After contact with skin, rinse immediately with plenty of water.
In case of accidental contact with eyes, rinse with plenty of clean water. In case of symptoms, seek medical attention.
Special precautions should be observed for pregnant women working with the drug when accidental self-injection is likely, as it may cause uterine contractions and decreased blood pressure in the womb.
Advice for veterinary practitioners: Medetomidine is a selective alpha-2 adrenoreceptor agonist. The following clinical effects may occur after absorption: dose-dependent sedation, respiratory depression, bradycardia, arterial hypotension, dry mouth and hyperglycemia, ventricular extrasystole. Respiratory and hemodynamic symptoms should be treated symptomatically.
Storage :
Dry, dark, out of reach of children at 40-77 F (5 °C to 25 °C).
Store vial in an outer box to protect it from light. Do not refrigerate or freeze.
The shelf life is 3 years.
After the first withdrawal from the bottle, the drug should be used within 28 days, if stored in a dark place at a temperature of 35-45F (2 ° C to 8 ° C).
For use in veterinary medicine!







Reviews
There are no reviews yet.